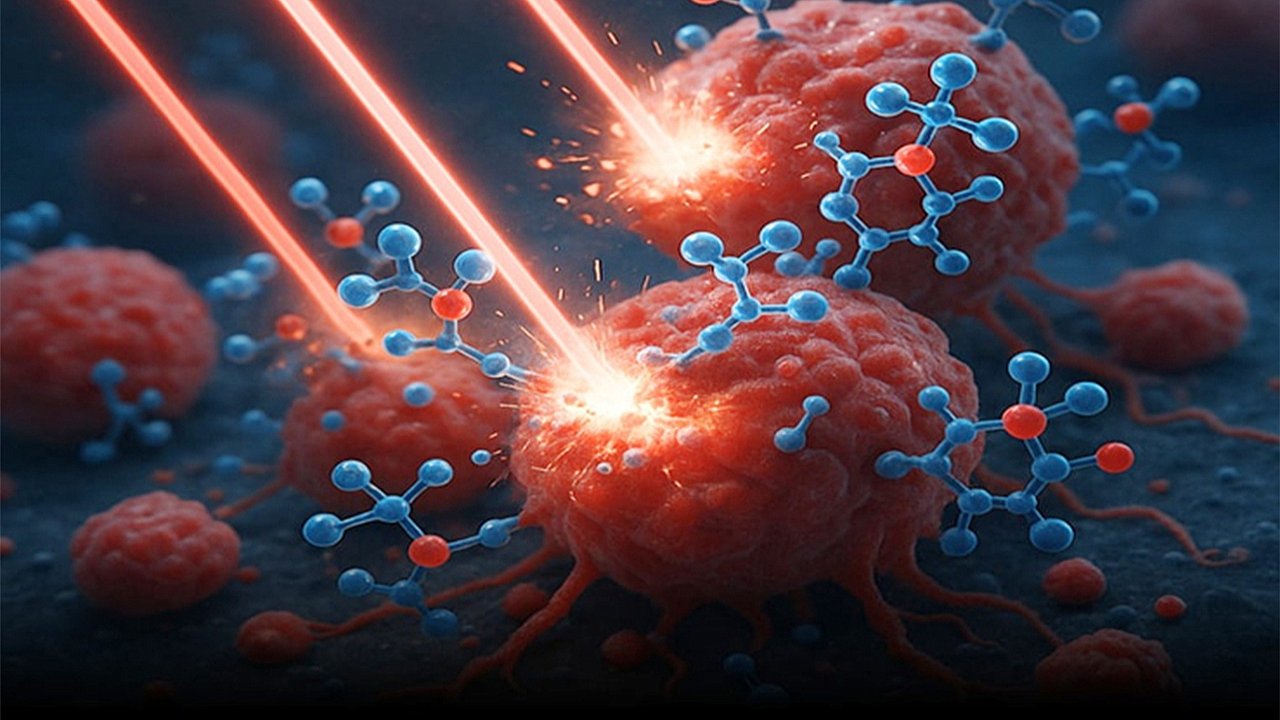
Molecular Jackhammers: A Groundbreaking Weapon Against Cancer Cells
Just as industrial jackhammers break through concrete, scientists have developed molecular jackhammers (MJHs)—nanoscale devices that mechanically destroy cancer cells by forcefully rupturing their membranes. Activated by near-infrared (NIR) light, these MJHs generate synchronized vibrations that deliver powerful mechanical blows at the molecular level, disrupting and ultimately killing the targeted cancer cells.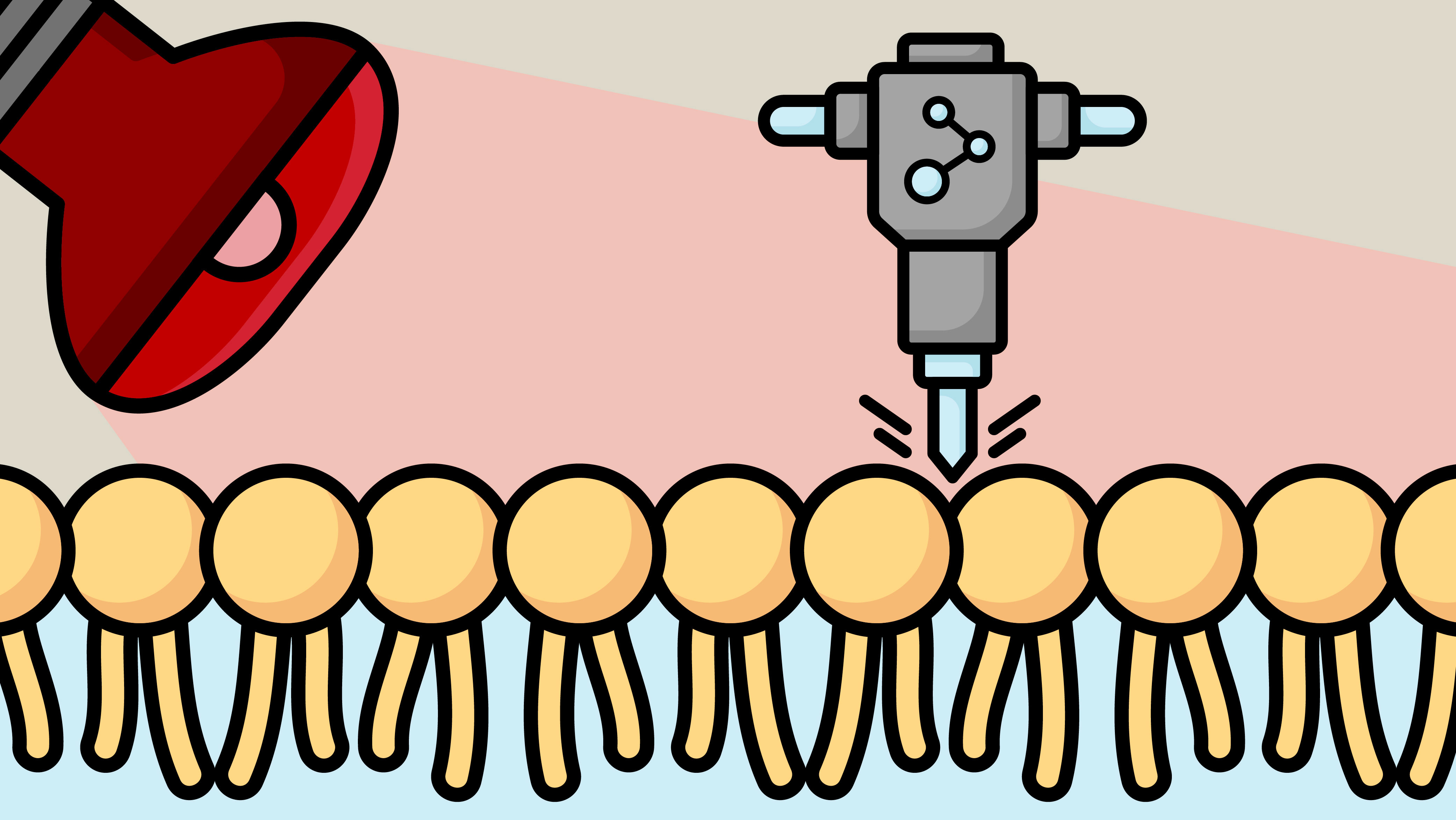 Image: Rachel Barton/Texas A&M EngineeringThis pioneering research was conducted by teams from Texas A&M University, Rice University, and the University of Texas-MD Anderson Cancer Center. The technique demonstrated remarkable results in both lab-grown melanoma cells and live mouse models. In vitro, the MJHs successfully killed 99% of melanoma cells. Even more impressively, half of the treated mice became entirely cancer-free following this therapy.How the Technology Works
Image: Rachel Barton/Texas A&M EngineeringThis pioneering research was conducted by teams from Texas A&M University, Rice University, and the University of Texas-MD Anderson Cancer Center. The technique demonstrated remarkable results in both lab-grown melanoma cells and live mouse models. In vitro, the MJHs successfully killed 99% of melanoma cells. Even more impressively, half of the treated mice became entirely cancer-free following this therapy.How the Technology Works
The key to this innovation lies in aminocyanine molecules, which form the basis of MJHs. These molecules carry a positive charge, allowing them to naturally attach to the negatively charged surface of cancer cells—specifically, the phospholipid bilayer of their membranes.Once bound to the cell membrane, the MJHs are activated by exposure to NIR light. This light falls just below the visible red spectrum and is invisible to the human eye. When irradiated, the MJH molecules undergo a process where their electrons are excited, producing plasmons. These plasmons induce intense molecular vibrations across the entire structure of the molecule.These rapid oscillations work like a barrage of microscopic hammer blows, violently striking the cell surface. The result is mechanical damage to the cell membrane, severe enough to cause decompensation and trigger cell death.A Safer, More Predictable Approach
Dr. Jorge Seminario, professor in the Artie McFerrin Department of Chemical Engineering at Texas A&M University, highlights a major advantage of the MJH method: its theoretical predictability. Using first-principles quantum chemistry calculations, researchers can simulate how the MJHs will perform before they’re ever tested in the lab.“This predictive power allows researchers to avoid the expensive and time-consuming trial-and-error processes that are common in drug development,” said Dr. Seminario. “It makes the entire technique more cost-effective and potentially safer.”Moreover, unlike chemotherapy or radiation, the mechanical action of MJHs is less likely to lead to treatment resistance. Cancer cells that typically adapt and survive chemical attacks have little defense against brute mechanical force. This dramatically lowers the chance of recurrence through resistance mechanisms.Future Potential and Customization
The researchers believe this is just the beginning for molecular jackhammers. The vast possibilities for molecular design could allow scientists to tailor the technology to specific cancer types or even individual patients.“This is one of the rare examples where a first-principles theoretical approach has been directly linked to experimental cancer treatment,” Dr. Seminario noted. “Typically, medical research avoids using quantum chemistry models despite their power to reveal atomic-level interactions.”Contributors to this groundbreaking study include Dr. Diego Galvez-Aranda from Texas A&M, Drs. Ciceron Ayala-Orozco and James M. Tour from Rice University, and Arnoldo Corona, Roberto Rangel, and Dr. Jeffrey N. Myers from the University of Texas-MD Anderson Cancer Center.As this research progresses, the hope is to move MJHs from lab models to clinical settings, offering a safer, more precise, and cost-effective tool in the fight against cancer.
Just as industrial jackhammers break through concrete, scientists have developed molecular jackhammers (MJHs)—nanoscale devices that mechanically destroy cancer cells by forcefully rupturing their membranes. Activated by near-infrared (NIR) light, these MJHs generate synchronized vibrations that deliver powerful mechanical blows at the molecular level, disrupting and ultimately killing the targeted cancer cells.
 Image: Rachel Barton/Texas A&M EngineeringThis pioneering research was conducted by teams from Texas A&M University, Rice University, and the University of Texas-MD Anderson Cancer Center. The technique demonstrated remarkable results in both lab-grown melanoma cells and live mouse models. In vitro, the MJHs successfully killed 99% of melanoma cells. Even more impressively, half of the treated mice became entirely cancer-free following this therapy.How the Technology Works
Image: Rachel Barton/Texas A&M EngineeringThis pioneering research was conducted by teams from Texas A&M University, Rice University, and the University of Texas-MD Anderson Cancer Center. The technique demonstrated remarkable results in both lab-grown melanoma cells and live mouse models. In vitro, the MJHs successfully killed 99% of melanoma cells. Even more impressively, half of the treated mice became entirely cancer-free following this therapy.How the Technology WorksThe key to this innovation lies in aminocyanine molecules, which form the basis of MJHs. These molecules carry a positive charge, allowing them to naturally attach to the negatively charged surface of cancer cells—specifically, the phospholipid bilayer of their membranes.Once bound to the cell membrane, the MJHs are activated by exposure to NIR light. This light falls just below the visible red spectrum and is invisible to the human eye. When irradiated, the MJH molecules undergo a process where their electrons are excited, producing plasmons. These plasmons induce intense molecular vibrations across the entire structure of the molecule.These rapid oscillations work like a barrage of microscopic hammer blows, violently striking the cell surface. The result is mechanical damage to the cell membrane, severe enough to cause decompensation and trigger cell death.A Safer, More Predictable Approach
Dr. Jorge Seminario, professor in the Artie McFerrin Department of Chemical Engineering at Texas A&M University, highlights a major advantage of the MJH method: its theoretical predictability. Using first-principles quantum chemistry calculations, researchers can simulate how the MJHs will perform before they’re ever tested in the lab.“This predictive power allows researchers to avoid the expensive and time-consuming trial-and-error processes that are common in drug development,” said Dr. Seminario. “It makes the entire technique more cost-effective and potentially safer.”Moreover, unlike chemotherapy or radiation, the mechanical action of MJHs is less likely to lead to treatment resistance. Cancer cells that typically adapt and survive chemical attacks have little defense against brute mechanical force. This dramatically lowers the chance of recurrence through resistance mechanisms.Future Potential and Customization
The researchers believe this is just the beginning for molecular jackhammers. The vast possibilities for molecular design could allow scientists to tailor the technology to specific cancer types or even individual patients.“This is one of the rare examples where a first-principles theoretical approach has been directly linked to experimental cancer treatment,” Dr. Seminario noted. “Typically, medical research avoids using quantum chemistry models despite their power to reveal atomic-level interactions.”Contributors to this groundbreaking study include Dr. Diego Galvez-Aranda from Texas A&M, Drs. Ciceron Ayala-Orozco and James M. Tour from Rice University, and Arnoldo Corona, Roberto Rangel, and Dr. Jeffrey N. Myers from the University of Texas-MD Anderson Cancer Center.As this research progresses, the hope is to move MJHs from lab models to clinical settings, offering a safer, more precise, and cost-effective tool in the fight against cancer.